Pharma Integration Platform: Connect SAP Veeva LIMS & Compliance Systems
March 24, 2026A pharma integration platform connects SAP S/4HANA, Veeva Vault, LIMS, MES, and regulatory databases into a single automated data flow, with full audit trails and role-based access controls that support 21 CFR Part 11 and EU GMP Annex 11 compliance. eZintegrations connects the complete pharmaceutical technology stack using 5,000+ pre-built API endpoints and Automation Hub templates, letting your IT team build the first integration in hours without custom code and without compromising the validation controls your QA team requires.
TL;DR
Pharmaceutical and biotech companies operate the most complex and compliance-constrained technology stacks in any industry. SAP for ERP, Veeva Vault for quality and regulatory, LIMS for laboratory data, MES for manufacturing execution, and TraceLink or SAP ATTP for serialisation. Most of these systems do not talk to each other in real time without a validated integration layer. The cost of disconnection in pharma is specific: batch release delays when LIMS results cannot reach SAP QM automatically, CAPA records in Veeva that do not propagate to ERP, regulatory submissions assembled manually from data held across six systems.
eZintegrations connects your pharma stack via 5,000+ pre-built API endpoints with the compliance controls that life sciences integration requires: full audit trails, immutable transaction logs, environment separation, and Human Approval Gates configurable by workflow type. If you are evaluating pharma integration platforms now, the system connection table, the step-by-step LIMS-to-SAP batch release workflow, and the Automation Hub template import link are all in this post.
The Problem: Pharma Systems That Cannot Talk to Each Other
Your LIMS just completed the release testing on Batch 2024-B-0447. The pH is within specification. The potency is confirmed. The microbiological results are clear. Every test in the batch record has passed.
Now someone needs to take those results from the LIMS, manually verify them, re-enter the key values into SAP QM, and initiate the usage decision workflow. That person is a QC analyst who also has eight other batches in various stages of testing. The batch sits in the queue until they get to it. Batch release, which could happen within minutes of the final test result, takes 4-8 hours on a good day and spills into the next business day when testing runs late.
This is not an unusual scenario. It is the operational baseline for most mid-size pharmaceutical manufacturers who have not yet connected their LIMS to their ERP.
The disconnection runs deeper than LIMS and SAP. In most pharma organisations, the following system pairs operate without automated data exchange:
- Veeva Vault Quality and SAP: A CAPA raised in Veeva does not automatically put the affected material on hold in SAP. Someone checks the CAPA, identifies the affected lot, and manually updates the batch status in SAP. If it is discovered during a weekend inspection, the manual step may not happen until Monday.
- LIMS and Veeva Vault QualityDocs: Stability study results in the LIMS are manually transcribed into Veeva documents for the Annual Product Quality Review. Every transcription is a data integrity risk.
- SAP and Veeva Vault RIM: Product registration data in Veeva RIM is not synchronised with the material master in SAP. When a regulatory variation updates an approved specification, the SAP QM inspection plan needs manual update.
- MES and SAP: Electronic batch records from the Manufacturing Execution System are not automatically reconciled with SAP production orders. Close-out requires manual comparison.
- TraceLink or SAP ATTP and WMS: Serialisation data from track-and-trace systems is not automatically available to the warehouse management system for real-time pack verification.
According to a 2025 industry survey cited by IntuitionLabs, integrating multiple applications is the number-one technology challenge for clinical operations teams in life sciences. By 2025, healthcare and life sciences accounted for over USD 2 billion in iPaaS spending, roughly 15% of the global iPaaS market, reflecting how urgent the integration problem has become in this sector.
The compliance dimension makes this more than an efficiency problem. Under 21 CFR Part 11, every electronic record that flows between systems must have a demonstrable, computer-generated audit trail. The FDA’s September 2025 final guidance on Computer Software Assurance (CSA) introduced a modern, risk-based approach to software validation, but the core requirement for traceable, integrity-protected data flows remains unchanged. When data is transferred manually, the audit trail is a paper log, not a system record.
Before vs After: Disconnected Pharma Stack vs Integrated Operations
| Dimension | Before eZintegrations | After eZintegrations |
|---|---|---|
| LIMS-to-SAP batch release | Manual transcription of test results from LIMS to SAP QM. 4–8 hour batch release delay per batch. | LIMS final result event triggers automated data push to SAP QM. Usage decision initiated within minutes. |
| CAPA propagation | CAPA raised in Veeva Vault Quality. Affected lot manually placed on hold in SAP by QA team. Hours to days depending on shift coverage. | Veeva Vault CAPA webhook triggers SAP material block via API. Affected lots on hold within minutes. Audit trail in both systems. |
| Stability study data | LIMS stability results manually transcribed into Veeva Vault documents for APQR. Each transcription is a data integrity risk. | LIMS stability result event triggers automated push to Veeva Vault document record. No manual transcription. |
| Regulatory variation updates | SAP QM inspection plan updated manually after Veeva RIM variation approval. Risk of testing against outdated specification. | Veeva RIM approval event triggers SAP QM inspection plan update via API. Specification always current. |
| Electronic batch record close-out | MES batch record manually compared to SAP production order for close-out. 2–4 hours per batch. | MES batch record completion triggers automated SAP production order reconciliation. Exceptions flagged. |
| Serialisation and pack verification | TraceLink or ATTP serial number data not available to WMS in real time. Manual pack verification step. | TraceLink event triggers real-time serial number push to WMS. Pack verification automated. |
| Audit trail for data transfers | Paper log or manual entry. Not computer-generated. Risk of 21 CFR Part 11 non-compliance. | Every integration transaction logged: timestamp, source system, destination system, payload hash, and outcome. Computer-generated audit trail. |
| Setup time for new integration | Custom API development: 8–16 weeks per connection. Requires GxP validation documentation. | Automation Hub template import: 2–8 hours configuration. Validation documentation templates available. |
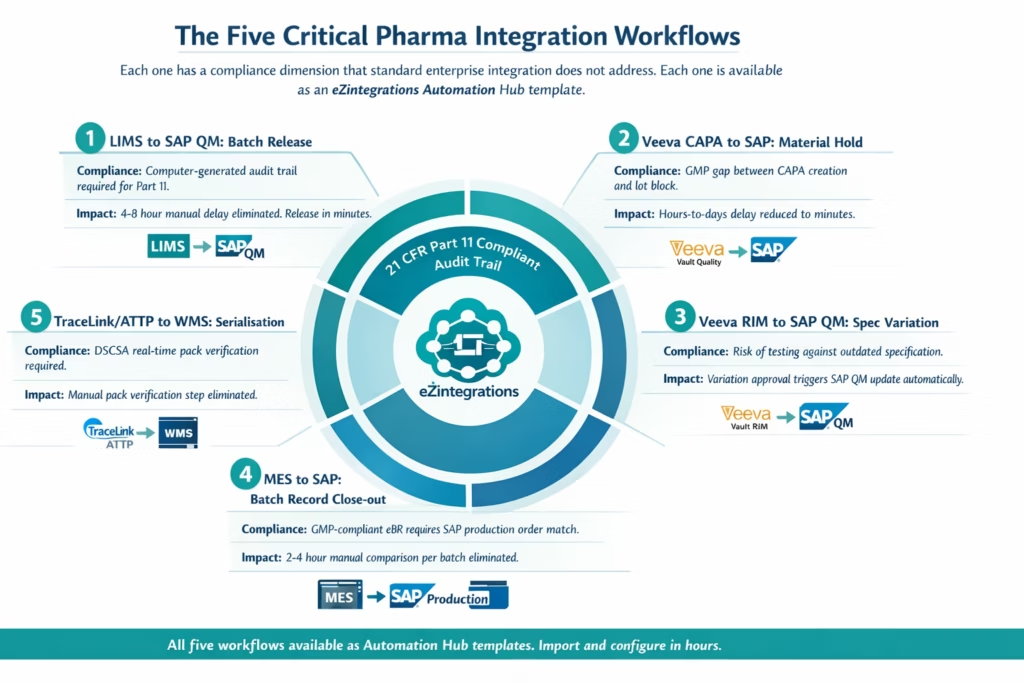
The Critical Pharma Integration Workflows
Pharmaceutical operations depend on five integration patterns more than any others. Each one has a compliance dimension that distinguishes pharma integration from standard enterprise integration. Here is what each workflow requires and why the integration layer must be built to handle it.
1. LIMS to SAP QM: Automated Batch Release
The most time-sensitive integration in pharmaceutical manufacturing. When final test results are recorded in the LIMS, the SAP QM usage decision workflow should fire within minutes, not hours. The integration requires: test result data mapped to SAP QM inspection lot fields, result status (pass/fail/conditional) translated to SAP usage decision codes, and the originating LIMS record referenced in the SAP audit trail.
For 21 CFR Part 11 compliance, the data transfer itself must be logged: which LIMS record was the source, when it was transferred, what values were mapped, and what the resulting SAP action was. Manual transcription cannot satisfy this requirement. An automated integration with an immutable transaction log can.
2. Veeva Vault Quality to SAP: CAPA and Material Hold Propagation
When a CAPA or deviation is raised in Veeva Vault Quality, any affected material in SAP must be placed on hold immediately. The delay between CAPA creation and SAP lot block is a GMP compliance gap. eZintegrations monitors Veeva Vault for CAPA events via webhook and triggers the SAP material block API call with the affected lot number and reason code. Both Veeva and SAP log the event with timestamps. The CAPA record in Veeva is updated with the SAP block confirmation reference.
3. Veeva Vault RIM to SAP QM: Regulatory Variation Propagation
When a regulatory variation is approved in Veeva Vault RIM, the updated specification must be reflected in the corresponding SAP QM inspection plan before the next batch is tested. Without integration, there is a window during which the SAP QM team is unaware that the approved specification has changed. The integration maps the Veeva RIM variation approval event to the specific SAP QM inspection plan and triggers the update, with the Veeva variation record number stored as the change reference in SAP.
4. MES to SAP: Electronic Batch Record Reconciliation
Manufacturing Execution System batch records must reconcile with SAP production orders for GMP-compliant electronic batch record close-out. The integration compares MES yield data, equipment usage, and operator records against the SAP production order, flags discrepancies for QA review, and initiates the SAP production order technical completion when reconciliation is clean. This workflow typically replaces a 2-4 hour manual comparison step per batch.
5. TraceLink or SAP ATTP to WMS: Serialisation and Pack Verification
Drug Supply Chain Security Act (DSCSA) compliance requires real-time serial number verification at the point of pack. The integration connects your serialisation system, either TraceLink or SAP Advanced Track and Trace for Pharmaceuticals, to your warehouse management system so that every pack-level transaction is verified against the commissioned serial number register in real time.
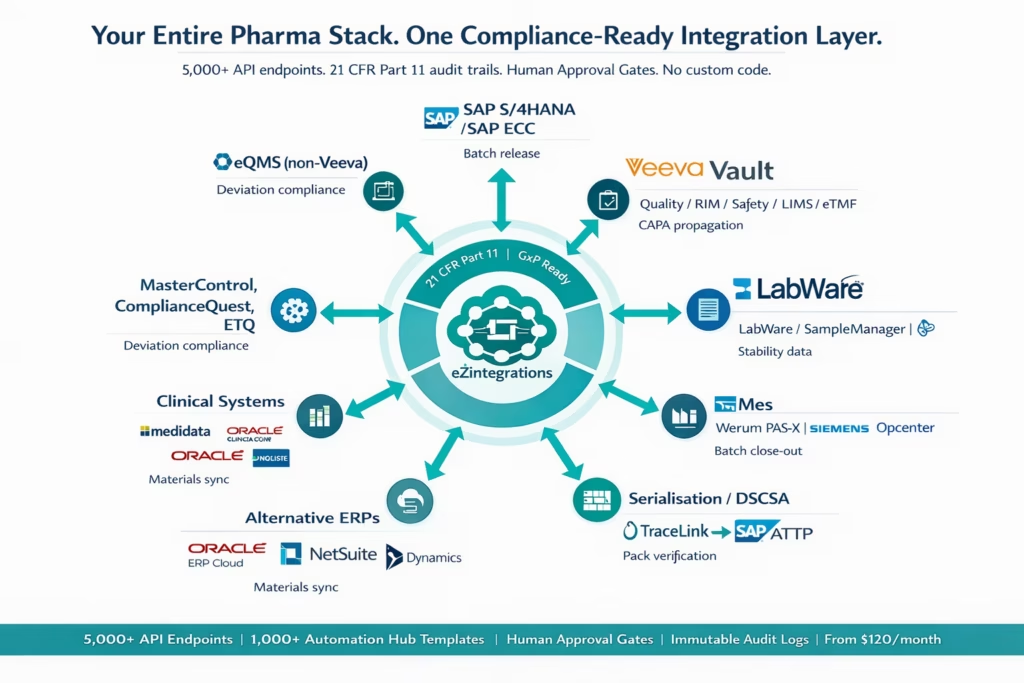
How eZintegrations Connects Your Pharma Stack
eZintegrations connects pharmaceutical and life sciences systems via an API catalog of 5,000+ endpoints. Every integration runs through the API catalog using REST and GraphQL connections. Systems not already in the catalog can be onboarded as self-service API or database connections without writing code.
For pharma-specific compliance requirements, the platform provides:
Full transaction audit trail: Every integration transaction is logged with timestamp, source system identifier, destination system identifier, payload (or payload hash for sensitive data), and outcome status. Logs are immutable and retained according to configurable retention policy. This is a computer-generated audit trail in the sense required by 21 CFR Part 11.
Human Approval Gates: Configurable human review steps for workflows where autonomous data transfer requires QA sign-off. For high-risk workflows (usage decision initiation, CAPA-triggered material holds, regulatory specification updates), a Human Approval Gate can require an authorised user to review and approve the data before the downstream API call fires.
Dev/Test/Production environment separation: Every integration is built and validated in Dev and Test before being promoted to Production. Environment separation is built into the platform architecture, supporting IQ/OQ/PQ-style validation approaches for integration workflows.
Role-based access control: Workflow configuration, credential management, and production promotion are restricted by role. Your integration developer and your QA validator have different permission sets.
Watcher Tools for compliance event monitoring: The Watcher Tool monitors source systems for specific events (CAPA status change in Veeva, test result completion in LIMS, batch record status change in MES) and triggers integration workflows automatically when the event occurs. No polling lag. No missed events.
Here is how the key pharma system categories connect:
ERP Systems: SAP S/4HANA, SAP ECC (via OData, BAPI, and SAP Integration Suite), Oracle ERP Cloud, NetSuite, Microsoft Dynamics 365 for Finance and Operations.
Quality and Regulatory: Veeva Vault Quality (QMS, QualityDocs, LIMS), Veeva Vault RIM, Veeva Vault Safety, MasterControl, ComplianceQuest, ETQ Reliance.
Laboratory: LabWare LIMS, Thermo Fisher SampleManager, Benchling, Dotmatics, Waters Empower CDS, and any LIMS exposing a REST API.
Manufacturing: Rockwell FactoryTalk MES, Siemens Opcenter, Werum PAS-X, GE Proficy, and any MES with a REST or database interface.
Serialisation: TraceLink, SAP Advanced Track and Trace for Pharmaceuticals (ATTP), Antares Vision.
Clinical: Medidata Rave, Veeva Vault eTMF, Oracle Clinical One, REDCap (via REST API).
The Automation Hub provides pre-built templates for the most common pharma integration patterns. Import a template, configure your system credentials and field mapping in the no-code canvas, validate in the Dev environment, and promote to production. The first integration goes live in hours.
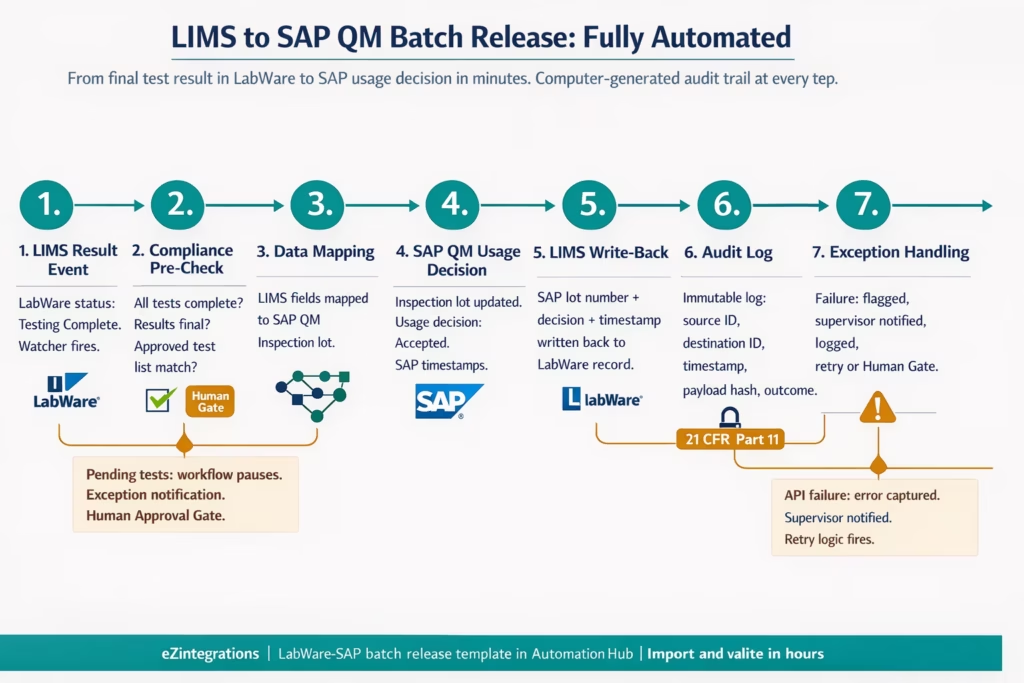
Step-by-Step: LIMS Test Results to SAP Batch Release
This is the core pharma integration workflow: the LIMS completes final release testing on a batch, and the SAP QM usage decision workflow fires automatically, with a full audit trail in both systems. The example runs on LabWare LIMS as the source and SAP S/4HANA QM as the destination. The integration runs on eZintegrations.
Step 1: LIMS Final Result Event (Watcher Tool) LabWare LIMS records the final test result for Batch 2024-B-0447. All results are within specification. The batch status in LIMS updates to “Testing Complete.” The eZintegrations Watcher Tool, monitoring the LabWare result status API for this status change, detects the event within seconds. The integration workflow triggers.
Step 2: Compliance Pre-Check (API Tool Call + Human Approval Gate) Before sending data to SAP, the workflow runs a pre-check: API Tool Call to LabWare: confirm all required tests are complete, no pending results, result status is final (not provisional) .Check against the approved test list for this product and batch type (retrieved from the Knowledge Base or a compliance rules database) . If any test is pending or provisional: workflow pauses. Exception notification sent to QC supervisor with full batch context. Human Approval Gate fires.
All pre-checks pass. Workflow continues.
Step 3: Data Transformation and Mapping (No-Code Canvas) The workflow reads the LIMS result record: batch number, product code, inspection lot reference, individual test results, units, reference ranges, and analyst ID. The no-code canvas maps each LIMS field to the corresponding SAP QM inspection lot field. The LabWare batch number maps to the SAP inspection lot number (or creates a cross-reference if the ID formats differ). The LIMS analyst ID maps to the SAP responsible user field.
Step 4: SAP QM Usage Decision Initiation (SAP API) The workflow calls the SAP QM usage decision API. The inspection lot is updated with the test results from LIMS. The usage decision is set to “Accepted” based on the result status mapping. The SAP system records the decision, timestamps it, and logs the originating external system (LabWare integration via eZintegrations).
Step 5: Write-Back to LIMS (LabWare API) The workflow writes back to LabWare: the SAP inspection lot number, the usage decision outcome, and the SAP timestamp are recorded in the LIMS batch record. This creates a bidirectional audit trail: LabWare shows that the SAP usage decision was initiated from this LIMS record, and SAP shows that the inspection lot data came from this LabWare result.
Step 6: Transaction Audit Log (Immutable) The eZintegrations platform logs the complete transaction: LabWare source record ID, SAP inspection lot number, timestamp of data transfer, field mapping applied, payload hash, outcome status (success or error), and the user ID under which the integration executed. This log is immutable and retained per the configured retention period. It is the computer-generated audit trail required under 21 CFR Part 11 for any automated data transfer between regulated systems.
Step 7: Exception Handling If the SAP API call fails (system unavailable, mapping error, validation error), the workflow does not silently fail. The error is captured, the LIMS record is flagged with the integration failure status, the QC supervisor receives an exception notification with full context (batch ID, error code, failed field mapping), and the transaction is logged as failed with the error detail. Retry logic fires for transient errors. Persistent failures require manual review via the Human Approval Gate.
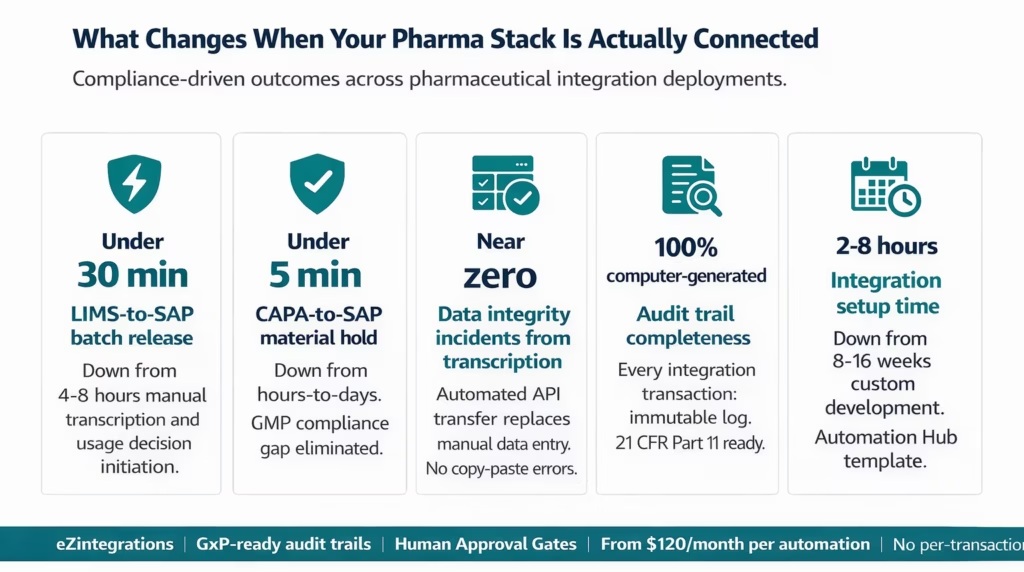
Key Outcomes and Results
The business case for pharma integration is measurable at every level: batch release speed, compliance audit readiness, data integrity incident reduction, and IT team capacity released from manual reconciliation work.
Batch release time: 4-8 hours reduced to under 30 minutes The LIMS-to-SAP integration eliminates the manual transcription and manual usage decision initiation step that creates the 4-8 hour gap between test completion and SAP batch release. Automated data transfer and usage decision initiation mean that a clean batch can move through release in under 30 minutes from final test record in LIMS.
CAPA-to-material hold propagation: Hours to under 5 minutes The Veeva CAPA-to-SAP material hold integration eliminates the period during which a CAPA exists in Veeva but the affected SAP lot is not yet blocked. This gap is a GMP compliance finding waiting to happen. With automated propagation, the lot is blocked in SAP within minutes of CAPA creation, and both systems carry the cross-reference.
Data integrity incidents from manual transcription: Reduced to near zero Manual transcription between LIMS and Veeva, or LIMS and SAP, is the primary source of data integrity incidents in many pharma IT environments. Automated integration eliminates the transcription step entirely. Data flows from source to destination system via an audited API call, not via a human reading a value and typing it somewhere else.
Audit trail completeness: 100% computer-generated Under 21 CFR Part 11, every electronic record and electronic signature must have a complete, computer-generated audit trail. Manual data transfers produce manual audit trails. Every eZintegrations transaction produces an automatic, timestamped, immutable log entry. Your next FDA inspection has complete, queryable transaction records for every automated data transfer.
Integration setup time: 8-16 weeks to 2-8 hours Custom pharma integration development, including requirements documentation, IQ/OQ/PQ validation scripting, and developer build time, typically takes 8-16 weeks per connection. eZintegrations Automation Hub templates reduce configuration to 2-8 hours. Validation documentation is not eliminated (the integration itself requires GxP validation), but the integration build time is, freeing validation effort for the testing and documentation phase rather than the development phase.
Healthcare and life sciences iPaaS: Fastest-growing vertical Healthcare and Life Sciences is the fastest-growing vertical in the global iPaaS market, growing at a 29.95% CAGR (Mordor Intelligence, 2026). Pharma organisations that invest in integration infrastructure now are building the data flow architecture that underpins AI-driven quality systems, automated regulatory submissions, and agentic compliance workflows in the years ahead.
How to Get Started
Pharma integration has a higher governance overhead than most enterprise integration projects, because every automated data flow between regulated systems requires validation evidence. The path below accounts for this. It is designed to get your first integration live in production, with appropriate validation documentation, faster than a custom development approach.
Step 1: Identify your highest-risk integration gap Use the five-workflow framework above. Which disconnection is creating the most compliance exposure or operational delay right now? For most pharmaceutical manufacturers, the answer is either the LIMS-to-SAP batch release gap (highest operational impact) or the Veeva CAPA-to-SAP material hold gap (highest compliance risk). Start with the one that your QA director or IT director identifies as highest priority.
Step 2: Import the relevant Automation Hub template Navigate to the Automation Hub and search for your system combination. The LabWare-to-SAP batch release template, the Veeva Vault CAPA-to-SAP material hold template, and the Veeva RIM-to-SAP QM variation sync template are available for import. The template includes the workflow structure, field mapping configuration, audit log settings, and exception handling logic pre-built.
Step 3: Configure credentials and map your fields in the Dev environment Add API credentials for your LIMS, ERP, and Veeva systems to the eZintegrations credential vault. Open the imported workflow in the no-code canvas. Review and adjust field mapping for your specific system configuration: SAP client number, inspection lot type, LIMS result status codes, and Veeva Vault document type identifiers. This step typically takes 2-8 hours.
Step 4: Validate in Dev and Test environments Run 20-50 test transactions in the Dev environment against real system data. Validate that: field mapping is correct, audit log entries are generated as expected, exception handling fires correctly for test error scenarios, and Human Approval Gate notifications reach the correct recipients. Document your test results. This documentation supports your IQ/OQ/PQ validation evidence for the integration workflow.
Step 5: Promote to Production with QA sign-off Promote the validated workflow to Production. In regulated pharma environments, production promotion requires QA review and authorisation of the validation evidence. eZintegrations Dev/Test/Prod environment separation ensures no configuration change reaches Production without a deliberate promotion step. Once in Production, the first automated data transfer runs, and the audit trail begins.
Ready to see it live? Book a free demo and bring your LIMS API documentation and your SAP QM configuration. We will build the batch release workflow in the session and show you the audit trail output.

Frequently Asked Questions
1. How do pharmaceutical companies use eZintegrations
Pharmaceutical companies use eZintegrations to connect ERP systems such as SAP S 4HANA SAP ECC Oracle ERP and NetSuite with quality and regulatory systems including Veeva Vault Quality RIM Safety and LIMS laboratory systems such as LabWare SampleManager Benchling and Dotmatics manufacturing execution systems including Werum PAS X Siemens Opcenter and Rockwell FactoryTalk serialisation systems such as TraceLink and SAP ATTP and clinical systems including Medidata and Oracle Clinical One. Common use cases include automated LIMS to ERP batch release CAPA to material hold propagation regulatory variation synchronisation electronic batch record reconciliation and DSCSA serialisation verification.
2. How long does it take to set up the first pharma integration
The integration configuration from Automation Hub template import through field mapping in the no code canvas typically takes two to eight hours. The validation phase required in GxP environments including IQ OQ and PQ testing adds additional time depending on organisational procedures and usually takes two to four weeks in a mid size pharma environment. This is significantly faster than custom integration development which typically takes eight to sixteen weeks because build time is eliminated and validation focuses on testing and documentation.
3. Does eZintegrations work with Veeva Vault SAP LabWare and Werum PAS X
Yes Veeva Vault connects through its REST API with OAuth 2 point 0 security covering Quality RIM Safety LIMS eTMF and QualityDocs. SAP S 4HANA and SAP ECC connect through OData and BAPI interfaces. LabWare LIMS connects through its REST API and Werum PAS X connects through its MES integration API. All are available in the eZintegrations API catalog and additional systems can be added through self service API onboarding without custom code.
4. How does eZintegrations handle 21 CFR Part 11 compliance for integration workflows
Every integration transaction generates an immutable audit log entry including timestamp source system identifier destination system identifier field mapping payload hash execution user ID and outcome status. This satisfies computer generated audit trail requirements under 21 CFR Part 11. Human approval gates can enforce electronic authorisation for high risk steps and Dev Test Production separation supports IQ OQ PQ validation models. Role based access control ensures only authorised users can modify production configurations.
5. Can eZintegrations integrate with on premises SAP deployments in pharma manufacturing
Yes eZintegrations is cloud native and connects to on premises SAP systems through exposed OData and BAPI interfaces. No on premises installation is required. Connectivity is typically established through secure API endpoint access with firewall whitelisting. This allows integration with existing SAP environments without infrastructure changes.
6. What validation documentation does eZintegrations provide for GxP environments
eZintegrations provides platform level documentation including system description architecture and security certifications. Automation Hub templates include pre built workflow documentation that supports IQ and OQ test script creation. Validation teams execute test scripts in the Test environment document outcomes and use immutable audit logs as evidence for validation and production promotion approval.
7. Does eZintegrations support the EU GMP Annex 11 requirements being updated in 2025 2026
Yes the updated EU GMP Annex 11 emphasises immutable audit trails multi factor authentication and proactive system security review. eZintegrations provides immutable audit logs that cannot be altered supports multi factor authentication for user access and enables configurable retention and export of integration logs for audit purposes ensuring compliance with evolving regulatory requirements.
Conclusion
The pharmaceutical technology stack is uniquely complex. Every system integration carries a compliance dimension that standard enterprise integration patterns do not address. Audit trails must be computer-generated. Data transfers between regulated systems must be validated. Human Approval Gates must be configurable for high-risk workflows. Environment separation is not optional.
eZintegrations was built to handle this level of compliance requirement without requiring custom development for every connection. Your LIMS connects to SAP QM. Your Veeva CAPA propagates to SAP material holds. Your MES batch records reconcile with SAP production orders. Your serialisation data reaches your WMS in real time. Each workflow has an immutable audit trail. Each workflow can be configured with Human Approval Gates where your QA procedures require them. Each workflow is validated in Dev and Test before it touches Production.
The first integration, from template import to validated production, takes hours rather than months. The compliance infrastructure is built into the platform rather than built separately for each connection.
Book a free demo and bring your LIMS API documentation and your SAP configuration. We will build the LIMS-to-SAP batch release workflow in the session and show you the audit trail output.
Or import the LIMS-to-SAP batch release template directly from the Automation Hub and run your first test transaction in the Dev environment today.